There are limited data on the long-term durability of transcatheter aortic valve replacement (TAVR) and surgery for severe aortic stenosis (AS) in patients of all risk levels. Management of patients with AS requires an understanding of bioprosthetic valve durability and failure. Revisit the evidence evaluating the 5-year incidence, outcomes, and predictors of hemodynamic structural valve disease in patients undergoing TAVR and surgery that will support best practices for treatment selection.
Disclosure of Conflicts of Interest
In accordance with the ACCME Standards for Integrity and Independence, Global Learning Collaborative (GLC) requires that individuals in a position to control the content of an educational activity disclose all relevant financial relationships with any ineligible company. GLC mitigates all conflicts of interest to ensure independence, objectivity, balance, and scientific rigor in all its educational programs.
Faculty:
Suzanne J. Baron, MD
Interventional Cardiologist
Lahey Hospital and Medical Center
Burlington, MAConsulting Fees: Abiomed, Biotronik, Boston Scientific Corp, Medtronic, Shockwave, Zoll
Research: Abiomed, Boston Scientific CorpJonathan A. Bradlow, MD, FACC
Cardiologist
Montefiore Medical Center
Bronx, NYNo relevant relationships reported
Kendra J. Grubb, MD
Surgical Director, Structural Heart & Valve Center
Emory University
Atlanta, GAConsulting Fees: Boston Scientific, Edwards, Medtronic
Research: Edwards Lifesciences, Medtronic
Reviewers/Content Planners/Authors:- Cindy Davidson has nothing to disclose.
- Howard Green has nothing to disclose.
- Colleen Resnick has nothing to disclose.
- Mara Siegel has nothing to disclose.
- Mario Trucillo, PhD, has nothing to disclose.
Learning Objectives
After participating in this educational activity, participants should be better able to:
- Evaluate the role and advantages of transcatheter aortic valve replacement (TAVR) and surgical aortic valve replacement (SAVR) therapeutic options based on recent lifetime management evidence
- Apply recent evidence on structural valve deterioration to validate the clinical use of TAVR in a variety of patient types
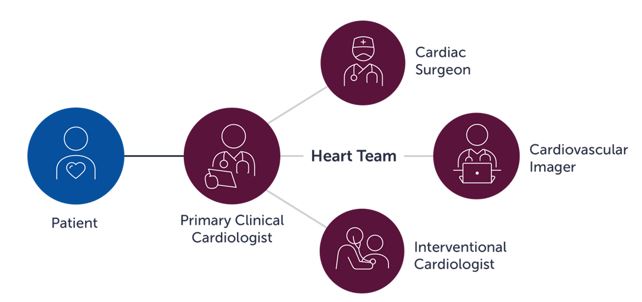
- Assess the value of the collaboration between interventional cardiologists and cardiac surgeons, the “heart team” approach, resulting in cooperative and improved care for patients
Target Audience
This activity is designed to meet the educational needs of cardiologists, cardiovascular surgeons, and interventional cardiologists.
Accreditation and Credit Designation Statements
 In support of improving patient care, Global Learning Collaborative (GLC) is jointly accredited by the Accreditation Council for Continuing Medical Education (ACCME), the Accreditation Council for Pharmacy Education (ACPE), and the American Nurses Credentialing Center (ANCC) to provide continuing education for the healthcare team.
In support of improving patient care, Global Learning Collaborative (GLC) is jointly accredited by the Accreditation Council for Continuing Medical Education (ACCME), the Accreditation Council for Pharmacy Education (ACPE), and the American Nurses Credentialing Center (ANCC) to provide continuing education for the healthcare team.Global Learning Collaborative (GLC) designates this enduring activity for a maximum of .25 AMA PRA Category 1 Credits™. Physicians should claim only the credit commensurate with the extent of their participation in the activity.
Global Learning Collaborative (GLC) designates this activity for .25 nursing contact hours. Nurses should claim only the credit commensurate with the extent of their participation in the activity.
 Global Learning Collaborative (GLC) has been authorized by the American Academy of PAs (AAPA) to award AAPA Category 1 CME credit for activities planned in accordance with AAPA CME Criteria. This activity is designated for .25 AAPA Category 1 CME credits. Approval is valid until October 25, 2023. PAs should claim only the credit commensurate with the extent of their participation in the activity.
Global Learning Collaborative (GLC) has been authorized by the American Academy of PAs (AAPA) to award AAPA Category 1 CME credit for activities planned in accordance with AAPA CME Criteria. This activity is designated for .25 AAPA Category 1 CME credits. Approval is valid until October 25, 2023. PAs should claim only the credit commensurate with the extent of their participation in the activity.Provider(s)/Educational Partner(s)

Our ultimate goal is to improve the care being delivered to patients, and our high quality, evidence-based CME initiatives reflect our dedication to the creation and execution of excellence and are the product of shared research, knowledge, and clinical practice skills across the healthcare continuum.
Commercial Support
This activity is supported by an independent educational grant from Medtronic.
Disclaimer
The views and opinions expressed in this educational activity are those of the faculty and do not necessarily represent the views of GLC and Medtelligence Education. This presentation is not intended to define an exclusive course of patient management; the participant should use his/her clinical judgment, knowledge, experience, and diagnostic skills in applying or adopting for professional use any of the information provided herein. Any procedures, medications, or other courses of diagnosis or treatment discussed or suggested in this activity should not be used by clinicians without evaluation of their patients’ conditions and possible contraindications or dangers in use, review of any applicable manufacturer’s product information, and comparison with recommendations of other authorities. Links to other sites may be provided as additional sources of information. Once you elect to link to a site outside of Medtelligence Education you are subject to the terms and conditions of use, including copyright and licensing restriction, of that site.
Reproduction Prohibited
Reproduction of this material is not permitted without written permission from the copyright owner.System Requirements
Our site requires a computer, tablet, or mobile device and a connection to the Internet. For best results, a high-speed Internet connection is recommended (DSL/Cable/Fibre). We also recommend using the latest version of your favorite browser to ensure compliance with W3C standards, such as Chrome, Safari, Firefox, or Microsoft Edge.
Publication Dates
Release Date:
Expiration Date:

















Facebook Comments